The Science of Iboga & Ibogaine
The science of ibogaine, from the forest to the clinic
Iboga has been used for thousands of years by the Bwiti people of Central Africa as a tool for healing, initiation, and ancestor connection. Modern science is now explaining what they always knew. This page is your guide — from the root to the receptor.
The Plant World
A Root with a 5,000-Year History
Tabernanthe iboga
Tabernanthe iboga is a slow-growing subshrub native to the rainforests of Central and West Africa — Gabon, Cameroon, the Republic of Congo, and Equatorial Guinea. It reaches one to two metres in height, with small yellow-orange flowers, and is unremarkable in appearance. It is the root bark — gathered, dried, and powdered — that contains the plant's extraordinary pharmacology. The alkaloids are concentrated in the bark of the root, not the leaves or stem, and harvesting is traditionally done with precision: only what is needed, and with ceremony.
Gabon · Cameroon · Republic of Congo · Equatorial Guinea

A National Treasure
In 2000, the Council of Ministers of the Republic of Gabon formally declared Tabernanthe iboga a national treasure and cultural heritage strategic reserve — one of the few instances in which a sovereign government has explicitly protected a psychoactive plant for its cultural and spiritual significance. The declaration placed iboga under the same legal framework as Gabon's natural heritage, restricting export and anchoring the plant's future to its country of origin.[13]
The Bwiti Tradition
The Bwiti religion of Central Africa is among the oldest continuous spiritual traditions on earth. It centres on the use of iboga — primarily the root bark — as a vehicle for healing, initiation, ancestor communication, and community repair. Two lineages are most significant: the Missoko (focused on healing and purification) and the Dissumba (focused on initiation and ancestral connection).
At the centre of every Bwiti ceremony is the Nganga — the healer and guide, trained over years or decades in the preparation and administration of iboga, the containment of the ceremonial space, and the interpretation of what arises. The Nganga does not observe from outside; they accompany the initiate through the experience.
Two modes of use exist within the tradition: the flood dose (sometimes called the full initiation dose) — a large, extended experience that can last 24 to 36 hours and is used for initiation and major transformation — and maintenance doses, smaller and more frequent, used for ongoing healing, spiritual practice, and community ritual.
The Bwiti initiation is understood as a rebirthing. The initiate symbolically dies — crossing from one state of being to another — and returns with new understanding, new identity, and a renewed relationship with their community and ancestors.
In the Bwiti cosmological frame, the visionary journey that iboga produces is not hallucination. It is a literal visit to the realm of the ancestors — an opportunity to receive wisdom, resolve inherited wounds, and return carrying that knowledge into life. This framing has been practised and refined over millennia. Modern neuroscience is only now beginning to understand why it works.
Voices of the Tradition
The Scholarly Record
The Molecular World
What's Actually in the Root
A Family of Compounds, Not a Single Molecule
Tabernanthe iboga root bark contains at least 12 identified alkaloids in the iboga structural family. This is not a simple plant with one active compound — it is a pharmacological ecosystem. Many practitioners and researchers believe the full alkaloid profile of the root bark produces effects that pure synthetic ibogaine alone cannot fully replicate, though this remains an active area of investigation.
The lead alkaloids are ibogaine, noribogaine, tabernanthine, ibogamine, and coronaridine — with ibogaine and its metabolite noribogaine being the most clinically studied.
Ibogaine — The Lead Compound
Ibogaine is the primary psychoactive alkaloid in Tabernanthe iboga root bark. It is responsible for the acute visionary experience and the majority of the receptor-binding activity that scientists have characterised. Once ingested, ibogaine is metabolised primarily by the CYP2D6 enzyme in the liver — converting it to noribogaine. People who are CYP2D6 poor metabolisers (a genetic variation affecting roughly 7–10% of the population) convert ibogaine more slowly, accumulating more of it in the bloodstream. This is why genetic screening before treatment is a standard part of responsible medical ibogaine protocols.
Ibogaine half-life: approximately 4–7 hours
Obach RS, Pablo J, Mash DC. Cytochrome P4502D6 catalyzes the O-demethylation of the psychoactive alkaloid ibogaine to 12-hydroxyibogamine. Drug Metab Dispos. 1998;26(8):764–768.[2]
Noribogaine — Where the Real Work Happens
Ibogaine is not, by itself, the whole story. When the liver processes ibogaine, the primary product is noribogaine — and noribogaine is not a passive metabolic byproduct. It is a pharmacologically active compound in its own right, and it behaves in ways that are fundamentally different from its parent molecule.
Noribogaine half-life: 28–49 hours — compared to ibogaine's 4–7 hours
After ibogaine has cleared the body, noribogaine persists in the bloodstream for days. The sustained anti-craving window that patients consistently report — dramatically reduced cravings for days or even weeks after a single treatment — appears to correlate directly with noribogaine plasma levels, not ibogaine.
Noribogaine is a G-protein-biased kappa-opioid receptor agonist: it binds kappa opioid receptors in a way that reduces craving without triggering the sedation and dysphoria that classic kappa agonists produce. This biased binding mechanism is one of the most significant pharmacological findings in ibogaine science.[3]
The following table summarises the major alkaloids identified in Tabernanthe iboga root bark, their approximate prevalence, and their primary known pharmacological action.
The Neural World
What Ibogaine Does to the Brain
Ibogaine does not fit into any single pharmacological category. It is simultaneously a serotonin reuptake stabiliser, an NMDA antagonist, an opioid receptor modulator, a nicotinic antagonist, and a neurotrophin amplifier. This pharmacological promiscuity — acting on many systems simultaneously — is likely why it produces effects that no single-target drug has been able to replicate.
Receptor Sites — Tap to Explore
Each site below represents a different way ibogaine interacts with the brain. Tap a receptor to see what it does in plain English.
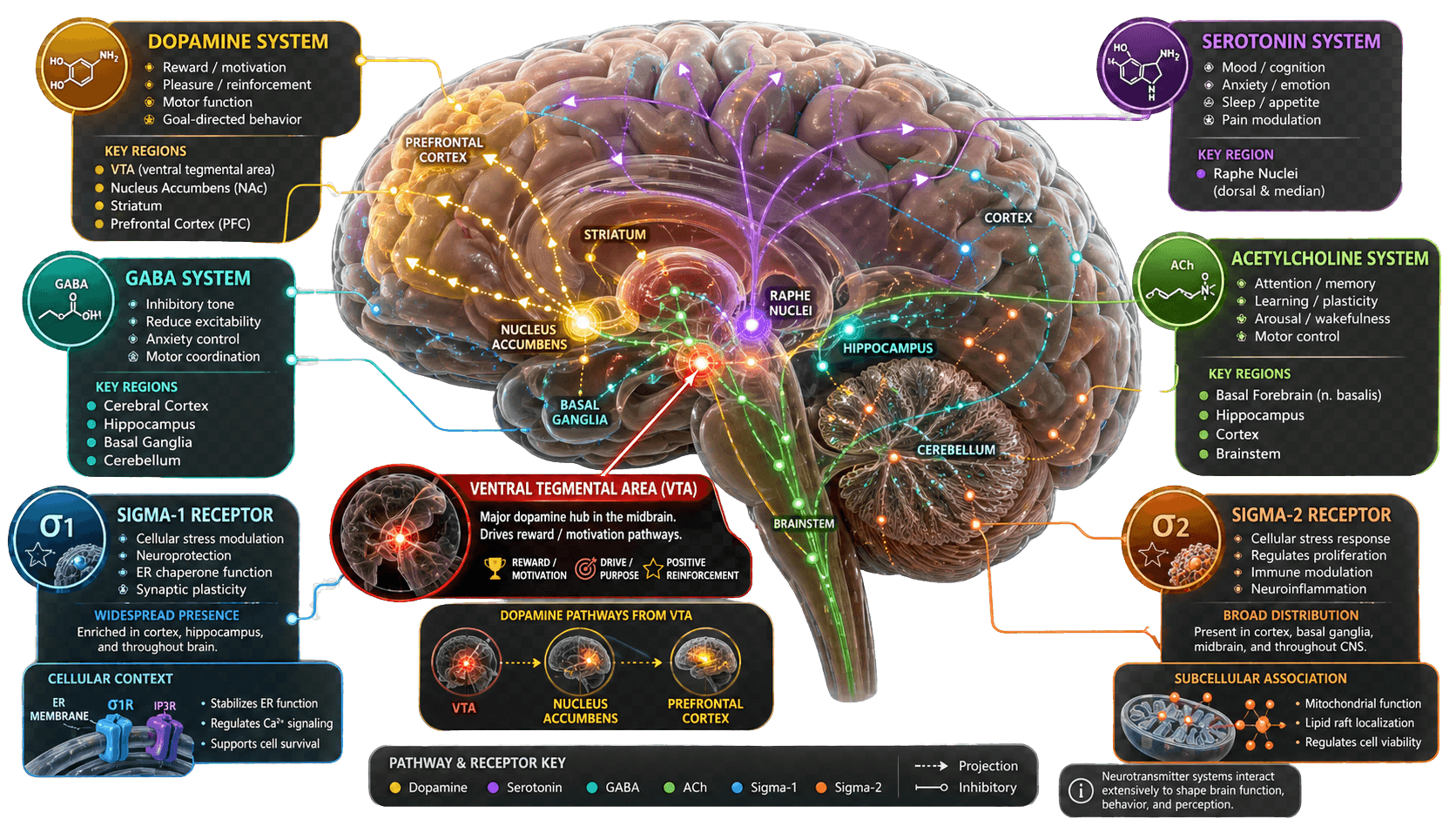
Click a system box on the diagram above, or tap a label below, to explore ibogaine's mechanism at that receptor site.
A Note on Cardiac Safety
Ibogaine also blocks hERG potassium channels in the heart — an effect that delays the heart's electrical repolarisation and can cause QT prolongation. This is why cardiac screening (ECG, electrolyte panel, medical history) and continuous ECG monitoring throughout treatment are non-negotiable requirements of any responsible ibogaine protocol. At Nekawa, ibogaine is administered inside a fully licensed hospital with ICU-level cardiac monitoring.[8]
Thurner P et al. Block of hERG channels by the indole alkaloid ibogaine. J Pharmacol Exp Ther. 2014;348(3):346–357.
The VTA — Resetting the Reward Circuit
The ventral tegmental area (VTA) is the origin of the brain's mesolimbic dopamine system — the reward pathway that addiction hijacks. VTA dopamine neurons project to the nucleus accumbens (the brain's primary reward centre) and to the prefrontal cortex.
In an addicted brain, this system becomes dysregulated. The 'allostatic set point' shifts: the brain requires drugs simply to feel normal, while natural rewards — food, connection, purpose — feel flat and motivationless. This is not a moral failure. It is a neurological state.
Ibogaine appears to temporarily suppress and then allow the resetting of VTA dopamine neuron firing patterns — not simply blocking or flooding the system, but enabling a recalibration toward the pre-addiction baseline. The mechanism is complex and still being characterised, but the clinical observation — dramatically reduced cravings in the days following treatment — is consistent across thousands of patients.
The Window of Wonder
One of ibogaine's most significant and least understood properties is what it does after the acute experience ends: it reopens a state of heightened brain plasticity that the brain normally only experiences during early childhood.
Neuroscientists call these 'critical periods' — windows of time when the brain is maximally malleable, when learning and rewiring happen at a pace impossible in the adult brain. Once these windows close during development, they were long thought to be closed permanently. Ibogaine appears to reopen them.
"Ibogaine, which produces this crazy long trip that in humans lasts three days, keeps the critical period in mice open for more than a month — and we haven't tested the closure."
"If you engineer these drugs to have a shorter, acute subjective effect, which is what a lot of companies are trying to do, you're going to get rid of the therapeutic efficacy of these drugs."
BDNF & GDNF — The Brain's Own Repair Proteins
During the Window of Wonder, the brain does not just become more plastic — it produces elevated levels of neurotrophic factors: BDNF (Brain-Derived Neurotrophic Factor) and GDNF (Glial Cell Line-Derived Neurotrophic Factor). These are the brain's own growth and repair proteins — the molecular signal for 'build new connections here.' Integration work done during this window — therapy, community, nature, movement — has the potential to be unusually impactful. This is why the weeks following ibogaine treatment matter as much as the treatment itself.
The Oneiric World
The Experience Inside
Ibogaine is classified by pharmacologists as an oneirogen — a compound that induces a waking dream state. This is not a metaphor. During an ibogaine session, the brain enters a prolonged state of intense, eyes-closed, autobiographical visionary experience unlike recreational intoxication, anaesthesia, or conventional psychedelic states.
What Happens During a Session
The typical progression: onset within 1–3 hours of ingestion; peak visionary experience from hours 4 to 8; a reflective, integrative phase from hours 8 to 20; residual sensitivity and afterglow for the following 24–48 hours. Throughout, the patient rests in a monitored room — ataxia (loss of coordination), sensitivity to sound and light, and the intensity of the inner experience make physical activity inadvisable and unnecessary. This is not something to be rushed.
The Bwiti Frame: Rebirthing, Rite of Passage, Ancestor Work
In the Bwiti tradition, the flood dose initiation is not understood as therapeutic in the Western clinical sense — it is a rite of passage. A crossing. The initiate is understood to symbolically die — to move from one state of being to another — and to return with new understanding, new identity, and a renewed relationship with community and ancestors. The Bwiti word for this transformation does not translate neatly into English. It describes a quality of having-been-somewhere-and-returned-changed.
In the Bwiti cosmological frame, the visionary experience is understood as a literal visit to the realm of the ancestors — those who have gone before and whose wisdom is available to the living if accessed through the right means. Iboga is that means. The Nganga's role is to guide the initiate through this territory safely, to interpret what arises, and to help integrate it into ongoing life.
What makes this framework remarkable — from a modern neuroscience perspective — is that it may be describing, in experiential terms, exactly what the neuroscience is showing in physiological terms. The brain during ibogaine is in a state of heightened plasticity, the Window of Wonder. The oneiric content of that state — the autobiographical review, the encounter with patterns, the sense of receiving understanding from somewhere outside ordinary thought — is the psychological material the open window allows to be processed and rewired. The Bwiti tradition has, over millennia, developed a ceremonial container precisely designed to make the most of that state.
Modern Interpretation
Contemporary therapy frameworks use the ibogaine experience as an extended period of introspective review. Patients frequently report watching their life's patterns — the roots of addiction, trauma, or depression — from a detached, observational perspective, as if reviewing a film of their own history. The emotional charge that ordinarily makes these memories unbearable is temporarily suspended, and the brain is sufficiently plastic to process and reframe them. This is not a cure. It is an opening — and what fills that opening, in the weeks of integration that follow, determines the depth of healing.
Nekawa's 28-day program is designed precisely to fill this window. The days before treatment prepare the nervous system; the days and weeks after — in nature, in community, with somatic support — are where the brain's extraordinary plasticity is given something worth wiring.
The Frontier
What We're Still Learning
The science of ibogaine is moving fast. What was speculative a decade ago is now peer-reviewed. What is speculative today will, in time, be established. This is what the frontier looks like.
The Open Questions
- →What determines the length of the Window of Wonder in individual humans — and can it be extended through integration practices?
- →Do whole-plant preparations of Tabernanthe iboga produce different effects than synthetic ibogaine, and if so, which alkaloids are responsible?
- →What are the long-term neuroplasticity effects across different populations, conditions, and treatment protocols?
- →Can the cardiac risk of ibogaine be reliably separated from the therapeutic mechanism — and what do drugs like Tabernanthalog (TBG) or other ibogaine analogues teach us?
- →What can the Bwiti tradition — with its millennia of empirical knowledge about preparation, ceremony, and integration — teach modern medicine about how to conduct these experiences responsibly?
You Are Part of This Story
Ibogaine research is one of the most active frontiers in neuroscience and psychiatry today. Veterans programmes. Stanford trials. MAPS protocols. The VA. Researchers at Johns Hopkins, Columbia, UC Berkeley, and dozens of other institutions are working to understand what the Bwiti people of Gabon have always known. The people seeking healing right now — at clinics like Nekawa — are participants in that frontier. Their outcomes, their recoveries, their willingness to step into something that mainstream medicine is only beginning to understand, is part of what builds the evidence base that will change treatment globally.
An Invitation
This is a story that is not finished. It is being written now, by scientists and by patients, by Nganga and by neuroscientists, by people who have tried everything else and found that ibogaine gave them something no other approach could: a window. What you do with the window is yours. We would like to help you open it.
References & Citations
- Nardou R, Sawyer E, Song YJ, et al. Psychedelics reopen the social reward learning critical period. Nature. 2023;618(7966):790–798. DOI ↗
- Obach RS, Pablo J, Mash DC. Cytochrome P4502D6 catalyzes the O-demethylation of the psychoactive alkaloid ibogaine to 12-hydroxyibogamine. Drug Metab Dispos. 1998;26(8):764–768. PubMed ↗
- Maillet EL, Milon N, Heghinian MD, et al. Noribogaine is a G-protein biased κ-opioid receptor agonist. Neuropharmacology. 2015;99:675–688. DOI ↗
- Jacobs MT, Zhang YW, Campbell SD, et al. Ibogaine, a noncompetitive inhibitor of serotonin transport, acts by stabilizing the cytoplasm-facing state of the transporter. J Biol Chem. 2007;282(40):29441–29447. DOI ↗
- Chen K, Kokate TG, Donevan SD, et al. Ibogaine block of the NMDA receptor: in vitro and in vivo studies. Neuropharmacology. 1996;35(4):423–431. DOI ↗
- Glick SD, Maisonneuve IS. Mechanisms of antiaddictive actions of ibogaine. Ann N Y Acad Sci. 1998;844:214–226. PubMed ↗
- Deecher DC, Teitler M, Soderlund DM, et al. Mechanisms of action of ibogaine and harmaline congeners based on radioligand binding studies. Brain Research. 1992;571(2):242–247. PubMed ↗
- Thurner P, Stary-Weinzinger A, Gafar H, et al. Mechanism of hERG channel block by the psychoactive indole alkaloid ibogaine. J Pharmacol Exp Ther. 2014;348(3):346–357. PubMed ↗
- Mash DC, Kovera CA, Pablo J, et al. Ibogaine: Complex pharmacokinetics, concerns for safety, and preliminary efficacy measures. Ann N Y Acad Sci. 2000;914:394–401. PubMed ↗
- Glue P, Neehoff S, Sabadel A, et al. Effects of ibogaine on opioid withdrawal in opioid dependent volunteers: a randomised, double-blind, placebo-controlled trial. J Psychopharmacol. 2020;34(9):910–919. DOI ↗
- Fernandez JW. Bwiti: An Ethnography of the Religious Imagination in Africa. Princeton University Press, 1982. Winner of the 1983 Herskovits Prize. Publisher ↗
- Samorini G. Studies on the iboga cults (series). Antrocom: Journal of Anthropology. 2024–2025. Open access ↗
- Council of Ministers of the Republic of Gabon. Ministerial decree declaring Tabernanthe iboga a national treasure and cultural heritage strategic reserve. 2000. Cited in: ICEERS. ICEERS ↗
Let’s connect.
No pressure — tell us a little about what you’re going through.